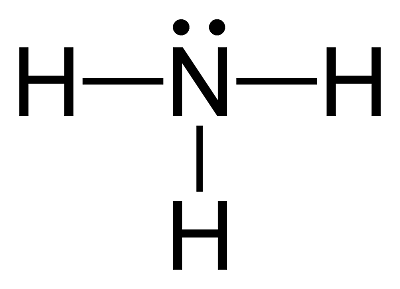
Ammonia Synthesis Production & Reaction | How Is Ammonia Made? - Video & Lesson Transcript | Study.com

Step-by-step guide to calculating unionized (toxic) ammonia. UIA of... | Download Scientific Diagram

Calculate the equilibrium constant (Kc) for the formation of NH3 in the following reaction: N2 (g) + 3H2 (g) 2NH3 (g) At equilibrium, the concentration of NH3, H2 and N2 are 1.2 ×
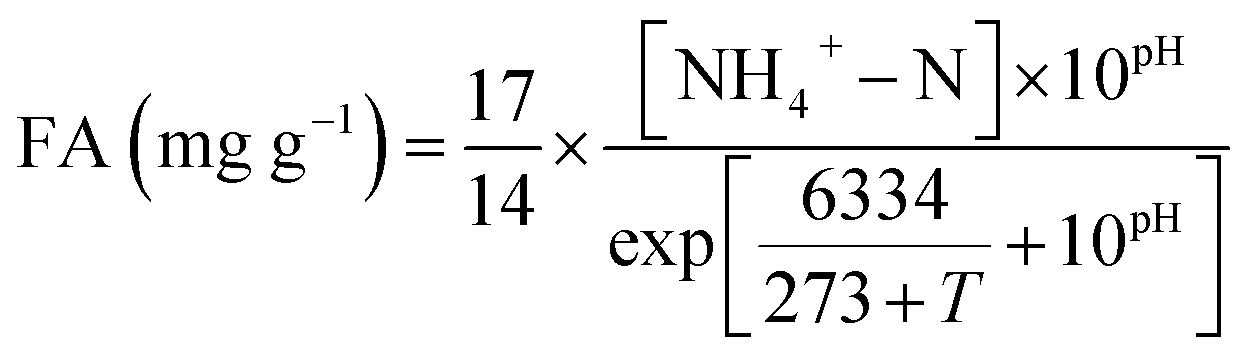
Characterizing the free ammonia exposure to the nutrients removal in activated sludge systems - RSC Advances (RSC Publishing) DOI:10.1039/C7RA10751J

SOLVED:The fertilizer, ammonium nitrate, is made by reacting ammonia with nitric acid. (Section 1.4) (a) Write a balanced equation, with state symbols, for the reaction of ammonia gas with nitric acid to

Calculate the pH of a 0.10 M ammonia solution . Calculate the pH after 50.0 mL of this solution is treated with 25.0 mL of 0.10 M HCl. The dissociation constant of ammonia, Kb=1.77xx10^(-5)

Question Video: Calculating the Equilibrium Constant for Concentration Given the Initial Amount of Each Reactant | Nagwa
How to calculate your Un-ionized ammonia levels Information below collected from: Francis-Floyd, Ruth, Craig Watson, Denise Pett



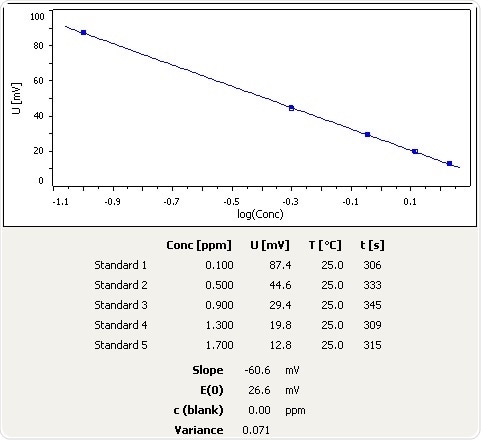
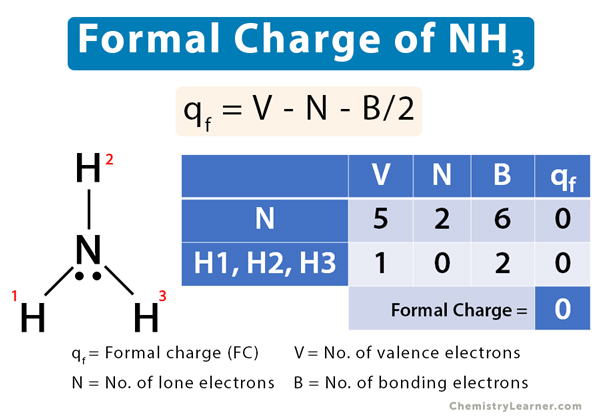
![Ammonia [NH3] Molecular Weight Calculation - Laboratory Notes Ammonia [NH3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/ammonia-molecular-weight-calculation-300x150.jpg)